Text source: European Commission
European Reference Networks (ERNs) are unique and innovative cross-border cooperation platforms between specialists for the diagnosis and treatment of rare or low prevalence complex diseases.
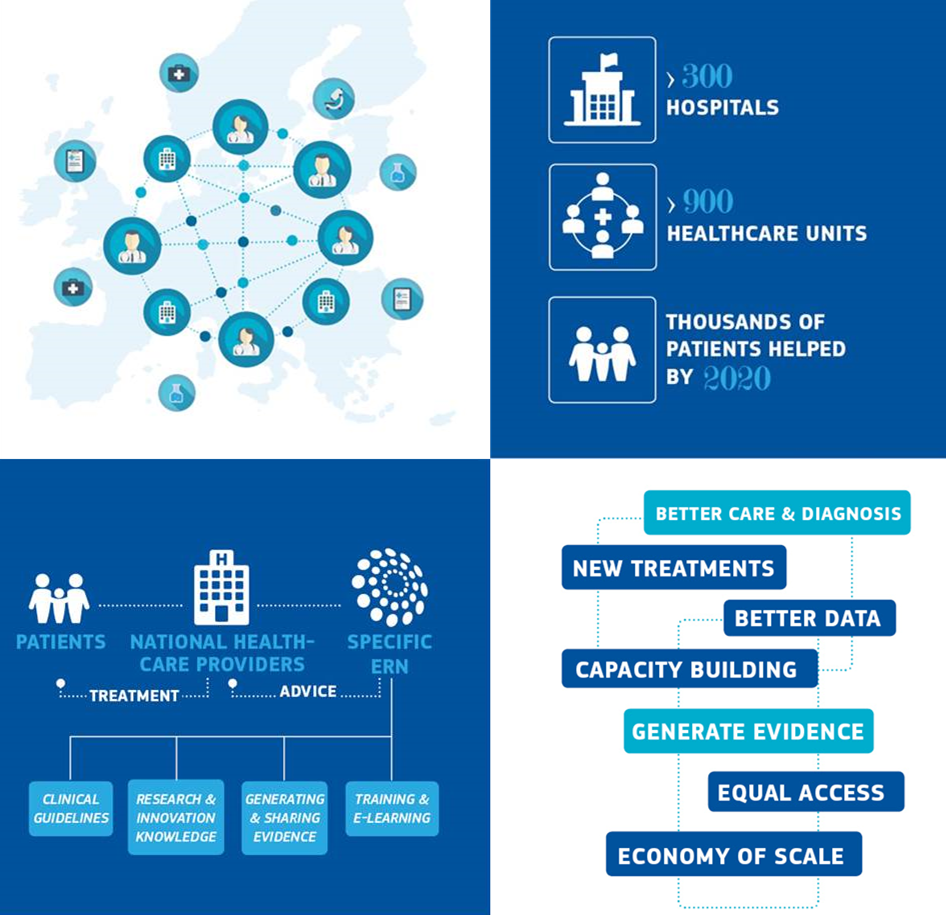
Overview
Health systems in the European Union aim to provide high-quality, cost-effective care. This is particularly difficult however, in cases of rare or low-prevalence complex diseases which affect the daily lives of around 30million EU citizens.
European Reference Networks (ERNs) are virtual networks involving healthcare providers across Europe. They aim to facilitate discussion on complex or rare diseases and conditions that require highly specialized treatment, and concentrated knowledge and resources.
How does it work?
To review a patient’s diagnosis and treatment, ERN coordinators convene ‘virtual’ advisory panels of medical specialists across different disciplines, using a dedicated IT platform and telemedicine tools.
The process and criteria for establishing an ERN and for selecting its members are set in EU legislation.
- Check ERN brochure for more information
24 Networks
The first ERNs were launched in March 2017, involving today more than 1600 highly-specialized healthcare units from 24 EU countries and Norway. 24 ERNs are working on a range of thematic issues including bone disorders, childhood cancer and immunodeficiency.
Legislation
- Directive 2011/24/EU on patients’ rights in cross-border healthcare
- Commission delegated decision (annex) defining the criteria and conditions that healthcare providers and the ERNs should fulfil
- Commission implementing decision (annex) defining criteria for establishing and evaluating ERNs
- Commission Implementing Decision (EU) 2019/1269 of 26 July 2019 amending Implementing Decision 2014/287/EU
The ERN initiative receives support from several EU funding programmes, including the Health Programme, the Connecting Europe Facility and Horizon 2020.
Communication material
- Information flyer for patients and healthcare professionals (all EU linguistic versions available)
- Flyer (all EU linguistic versions available)
- Brochure (all EU linguistic versions available – Norwegian version)
- Video clips:
- Rare diseases: how the European Reference Networks support patients
- Rare diseases: how the European Reference Networks support health professionals
- ERN featured in the « EU protects » campaign, how the EU connected experts to treat epilepsy
- Information video for patients and healthcare professionals (available with subtitles in all EU languages and Norwegian)
- The stories of Elisa, Paula, Jasper and Daniel (available with subtitles in all EU languages and Norwegian)
- Presentation of the ERNs (Video clip available in all EU languages and Norwegian, with or without subtitles)

