What about Rare Eye Diseases (RED)?
- Rare Eye Diseases (RED) are the leading cause of visual impairment and blindness for children and young adults in Europe
- There are more than 900 REDs
- From the most prevalent diseases such as retinitis pigmentosa (estimated prevalence of 1 in 5 000) to some very rare entities described only once or twice in medical literature
What is ERN-EYE?
ERN-EYE is a European reference network dedicated to Rare Eye Diseases.
ERN-EYE covers 24 out of 27 EU countries, with 52 full members and 8 affiliated partners, across European Union to guarantee the best coverage of more than 900 Rare Eye Diseases.
The coordinator is Pr Hélène Dollfus, Hôpitaux Universitaires de Strasbourg, France.
All members were approved by their national authority and positevely assessed by an independent body.
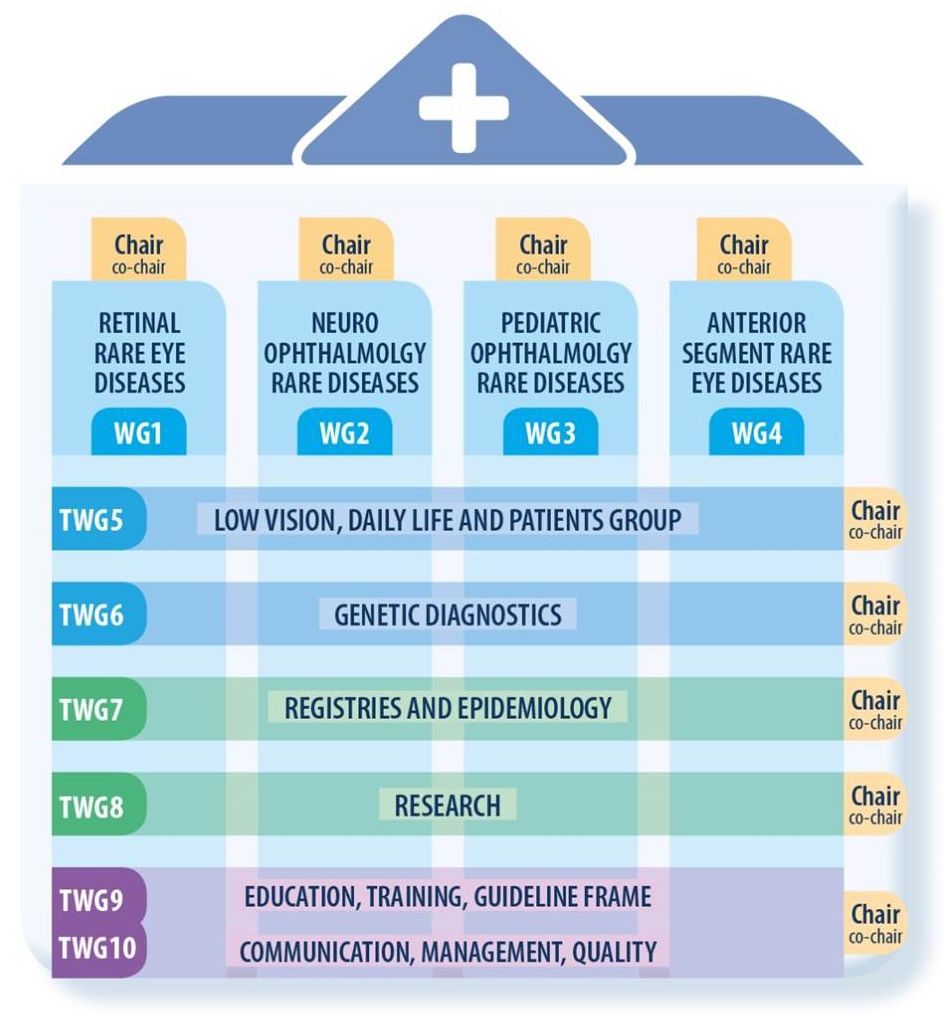
There are 4 working groups (WG) and 6 transversal working groups (TWG)
 WG1: Retinal Rare Eye Diseases WG1: Retinal Rare Eye Diseases |  TWG6: Genetic Diagnostics TWG6: Genetic Diagnostics |
 WG2: Neuro-Ophthalmology Rare Diseases WG2: Neuro-Ophthalmology Rare Diseases |  TWG7: Registries & Epidemiology TWG7: Registries & Epidemiology |
 WG3: Pediatric Ophthalmology Rare Diseases WG3: Pediatric Ophthalmology Rare Diseases |  TWG8: Research TWG8: Research |
 WG4: Anterior Segment Rare Eye Diseases WG4: Anterior Segment Rare Eye Diseases |  TWG9: National Integration TWG9: National Integration |
 TWG5: Low Vision, Daily Life and Patients Groups TWG5: Low Vision, Daily Life and Patients Groups |  TWG10: CPMS & Digital Medecine TWG10: CPMS & Digital Medecine |
52 full members & 8 affiliated partners
What are ERN-EYE’s Goals?
- Improve patient trajectory in the EU : Virtual clinic
- Reduce inequalities of patient care in the EU
- Reduce the time to diagnosis and care
- Ensure molecular diagnosis to the largest number of patients in the EU
- Bring diagnosis to the undiagnosed
- Facilitate involvement of patients in clinical trials
- Give access to innovation
- Provide tools for education and communication
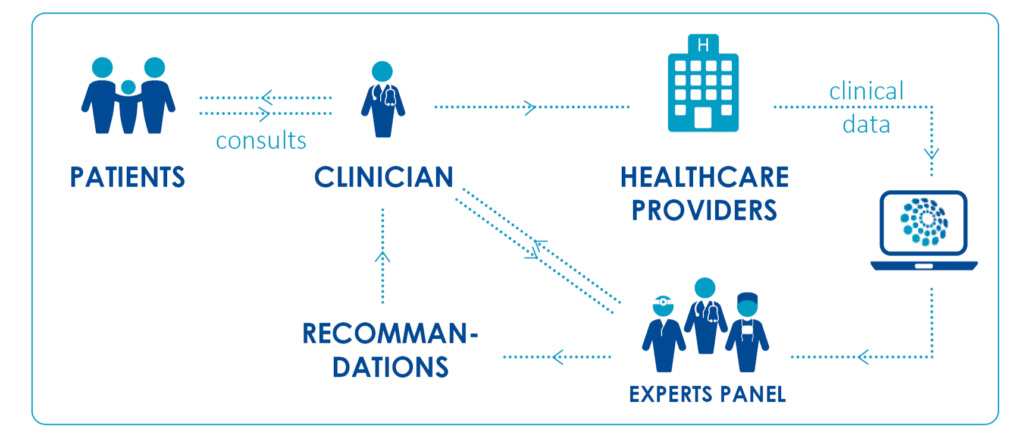
What is the clinical virtual care?
The Network main aim is the development of a virtual clinic to facilitate cross-border dissemination of expertise. The Clinical Patient Management System (CPMS) was launched by the European Commission (DG SANTE) in November 2017. It’s a web-based application where healthcare professionals from the European Reference Networks (ERNs) can discuss real patient cases. This platform aims at supporting ERNs in improving the diagnosis and treatment of rare or low prevalence complex diseases across national borders of Member States in Europe.
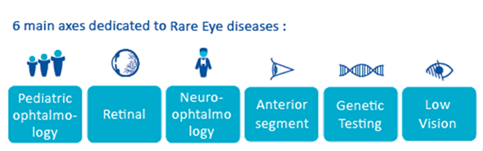
What are ERN-EYE’s working topics?
4 clinical thematic working groups
- Working group 1: Retinal rare eye diseases
- Working group 2: Neuro-ophthalmology rare diseases
- Working group 3: Paediatric ophthalmology rare diseases
- Working group 4: Anterior segment rare eye diseases
6 transversal working groups
- Transversal Working group 5: Low vision, daily life and patient group
- Transversal Working group 6: Genetic diagnostics
- Transversal Working group 7: Registries & epidemiology
- Transversal Working group 8: Research
- Transversal Working group 9: National Integration
- Transversal Working group 10: CPMS & Digital Medecine

